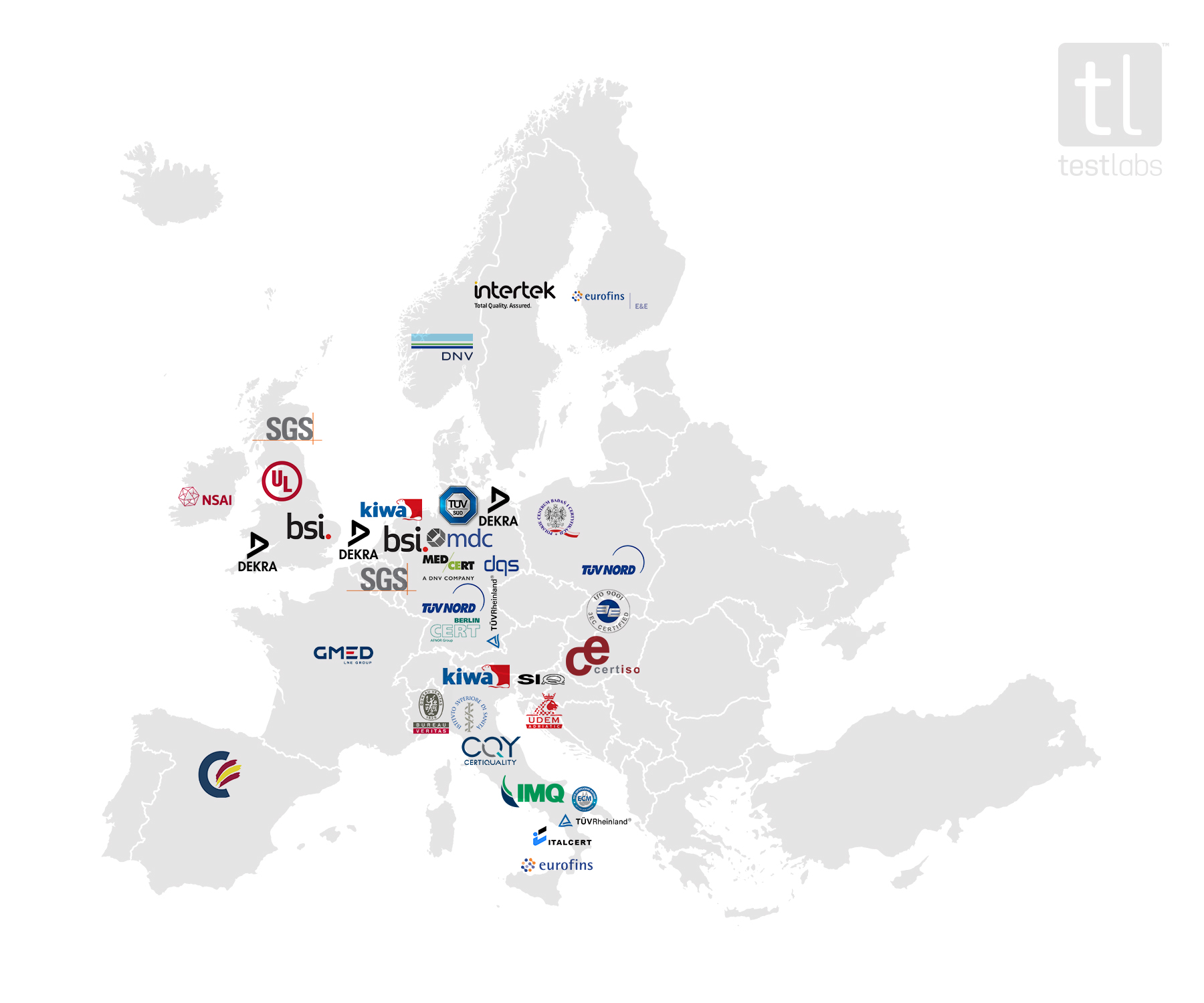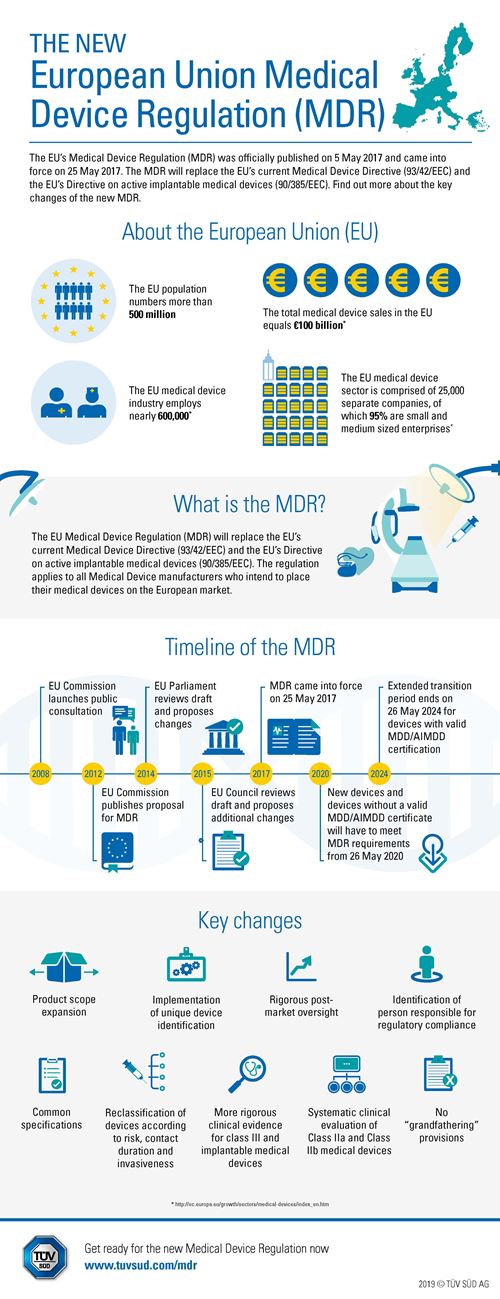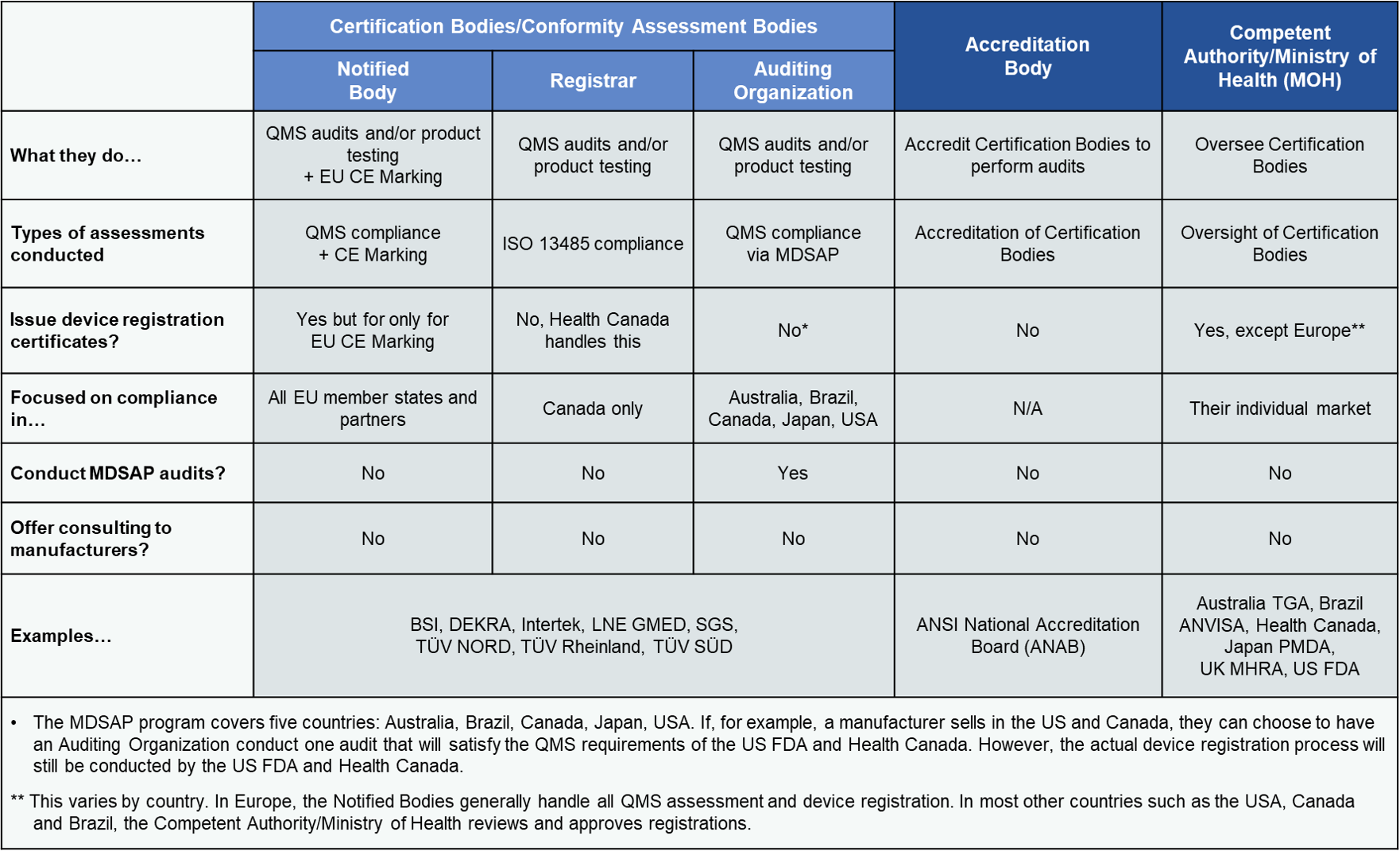
Auditing Organization (AO) versus Notified Body (NB) versus Registrar. What's the difference? – Oriel STAT A MATRIX Blog

BVMed on X: "For today's "Day of Application" of the EU Medical Device Regulation #MDR, we offer various infographics on the timeline, certificates, building sites and Notified Bodies. https://t.co/Eu9Am4ZCWy #MDReady #Medtech https://t.co/1eWZKhouhr" /
![Requirements Relating to Notified Bodies for EU MDR [Video] - LearnGxP: Accredited Online Life Science Training Courses Requirements Relating to Notified Bodies for EU MDR [Video] - LearnGxP: Accredited Online Life Science Training Courses](https://learngxp.com/wp-content/uploads/2021/04/ELM-320-01-Requirements-Relating-to-Notified-Bodies-for-EU-MDR.png)
Requirements Relating to Notified Bodies for EU MDR [Video] - LearnGxP: Accredited Online Life Science Training Courses

How will the changing role of Notified Bodies after the MDR and IVDR affect your company? | Proclinical Blogs
![ARTICLE] Why You Need To Review Your Technical Documentation NOW (And 8 Pitfalls to Avoid at all Costs) - Medidee Services ARTICLE] Why You Need To Review Your Technical Documentation NOW (And 8 Pitfalls to Avoid at all Costs) - Medidee Services](https://medidee.com/wp-content/uploads/2022/08/Technical-Documentation-Infographic.png)
ARTICLE] Why You Need To Review Your Technical Documentation NOW (And 8 Pitfalls to Avoid at all Costs) - Medidee Services
Availability and capacity of notified bodies to carry out conformity assessments for COVID-19 related medical devices and in vit

Designation process of MDR/IVDR Notified Bodies - update · MDlaw – Information platform on European medical device regulations